TRTpred, a sensitive in silico predictor of tumor reactive clonotypes. Credit: Nature Biotechnology (2024). DOI: 10.1038/s41587-024-02232-0
× Close
TRTpred, a sensitive in silico predictor of tumor reactive clonotypes. Credit: Nature Biotechnology (2024). DOI: 10.1038/s41587-024-02232-0
Using artificial intelligence, Ludwig Cancer Research scientists have developed a powerful predictive model to identify the most potent cancer-killing immune cells for use in cancer immunotherapies.
The predictive model, combined with additional algorithms, is described in the current issue of the journal. Nature Biotechnologycan be applied to personalized cancer treatment that tailors therapy to the unique cellular makeup of each patient's tumor.
“The implementation of artificial intelligence in cellular therapy is new and could be a game-changer, offering patients new medical options,” said Alexandre Harari of Ludwig-Lausanne, who led the study with graduate student Rémy Petrimand. of
Cellular immunotherapy involves extracting immune cells from a patient's tumor, optionally engineering them to enhance their natural cancer-fighting abilities, and reintroducing them back into the body after expansion in culture. T cells are one of two main types of white blood cells, or lymphocytes, that circulate in the blood and patrol for virus-infected or cancerous cells.
T cells that infiltrate solid tumors are called tumor-infiltrating lymphocytes, or TILs. However, not all TILs are effective in recognizing and attacking tumor cells.
“Only a fraction actually have a tumor reaction—the majority do,” Harari explained. “The challenge we set for ourselves was to identify the few TILs that are equipped with T-cell receptors that can recognize antigens on the tumor.”
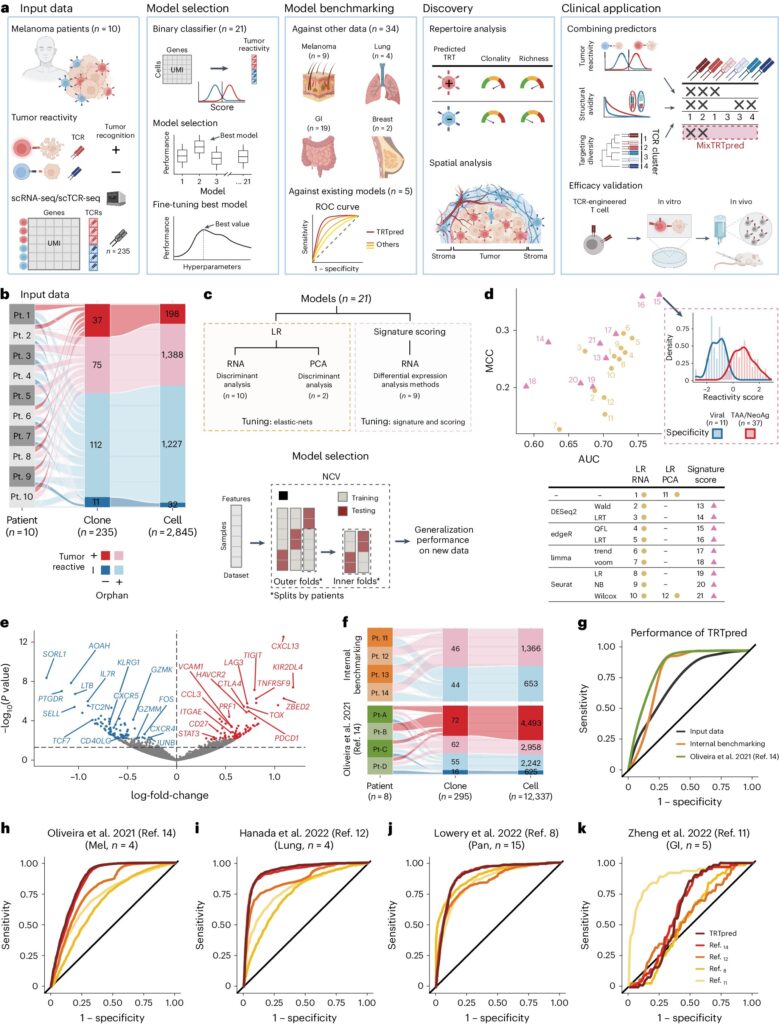
To do this, Harari and his team developed a new AI-powered predictive model, called TRTpred, that can classify T-cell receptors (TCRs) based on their tumor response. Is. To develop TRTpred, they used 235 TCRs collected from patients with metastatic melanoma, the tumors already classified as reactive or non-reactive.
The team loaded global gene expression — or transcriptomic — profiles of T cells that carry each TCR into a machine learning model to identify patterns that distinguish tumor-reactive T cells from inactive counterparts. do
“TRTpred can learn from one T cell population and create a rule that can be applied to a new population,” Harari explained. “Therefore, when a new TCR is encountered, the model can read its transcriptomic profile and predict whether it is tumor reactive or not.”
The TRTpred model analyzed TILs from 42 patients with melanoma and gastric, lung, and breast cancer and identified tumor-reactive TCRs with approximately 90% accuracy. The researchers further refined their TIL selection process by applying a secondary algorithmic filter to screen for only those tumor-reactive T cells with “high avidity”—that is, those that bind strongly to tumor antigens.
“TRTpred is specifically a predictor of whether the TCR is tumor reactive or not,” Harari explained. “But some tumor-reactive TCRs bind very strongly to tumor cells and are therefore very effective, while others do so only weakly. Distinguishing strong binders from weak ones would translate into efficacy. Is.”
The researchers demonstrated that T cells flagged by both the TRTpred and secondary algorithms are tumor reactive and more avidly found embedded within the tumor rather than in the adjacent supporting tissue, known as the stroma. This finding is consistent with other research showing that effector T cells typically infiltrate deep into tumor islets.
The team then introduced a third filter to better identify different tumor antigens. “What we want is for the TILs to target as many different antigens as possible,” Harari said.
This final filter sorts TCRs into groups based on similar physical and chemical properties. The researchers hypothesized that the TCRs in each cluster recognize the same antigen. “Therefore, we amplify one TCR from within each cluster, so that we can maximize the probability of distinct antigen targets,” said Vincent Zotte, a computational scientist at Ludwig-Lausanne who studied TCR avidity and TCR clustering. Developed the algorithm.
The researchers call the combination of TRTpred and algorithmic filters MixTRTpred.
To validate their approach, Harari's team cultivated human tumors in mice, extracted TCRs from their TILs, and used the MixTRTpred system to identify T cells that were tumor-reactive. I had high affinity and targeted multiple tumor antigens. They then engineered T cells from mice to express these TCRs and showed that these cells could eradicate tumors when transferred into mice.
“This approach promises to overcome some of the shortcomings of current TIL-based therapies, especially for patients dealing with tumors that are currently unresponsive to such therapies,” said Georg Kokos, director of Ludwig Lausanne. said, a co-author of the study who is planning to launch a follow-up study. A Phase I clinical trial that will test the technology in patients.
“Our combined efforts will lead to an entirely new type of T-cell therapy.”
More information:
Rémy Pétremand et al, Identification of clinically relevant T cell receptors for personalized T cell therapy using a combinatorial algorithm, Nature Biotechnology (2024). DOI: 10.1038/s41587-024-02232-0
Journal Information:
Nature Biotechnology